Many of my articles here (...ok most…) are deep dives, because the subject matter is often complex. In addition, I often write about topics that have been misconstrued by medicine in general, or there is confusion courtesy of fear mongering from functional medicine providers or naturopaths, or a story or article has been mishandled by the press. And yes, sometimes it’s an ungodly combination of all of the above. In these cases I want you to have detailed background information and want to walk you through all the points step-by-step so you can be prepared to advocate for yourself and know who you should ignore and even block on social media.
However, I also frequently come across medical articles that I find interesting, and I think, “I should write about that,” but when I sit down at my computer, it turns out I don’t have that much to say. The article typically provides a really interesting point or two, but not enough for a full post. Nothing controversial, just a “Well, I’m glad I know that because now I am more equipped to advise my patients.” Then this week I thought, “Why don’t I combine several of those articles into one post?” And that’s what we’re going to do today and cover:
Hysterectomy rates after endometrial ablation
Effectiveness of hormonal IUD at 8 years for contraception and stopping periods
Health ramifications of removing your ovaries before age 65?
So let’s get started!
How Effective is an Endometrial Ablation?
An endometrial ablation is a procedure where the lining of the uterus (the endometrium) is essentially burned away to treat heavy periods. There are a variety of ways this can be accomplished, but what we’re going to focus on here is the effectiveness, or one measure of effectiveness– whether or not the procedure is ultimately followed by a subsequent hysterectomy.
There are four main treatment options for heavy bleeding (and here I’m assuming there are no fibroids or bleeding disorders): hormones, non hormonal medications (typically nonsteroidal anti-inflammatory drugs and tranexamic acid), endometrial ablation, and hysterectomy. In general, the hormonal IUD and endometrial ablation are considered to be about on par in terms of effectiveness and the most effective non-hysterectomy options.
The paper that caught my eye is a meta analysis or a review of over 50 studies specifically looking at the hysterectomy rate, which is a pretty good proxy for “this procedure really did not work well enough for me.” The researchers found that at one year after an endometrial ablation about 4.3% of people had a hysterectomy and by 5 years that rate had risen to 12.3%. Interestingly, this rate was pretty consistent across the different studies that were included in the analysis, suggesting this is a pretty good number to use. Only two studies had 10 year follow up data, and they were relatively small so it is hard to be firm about the conclusion, but the hysterectomy rates at 10 years after an ablation ranged from 17.6% to 34.4%.
This paper can’t tell us who was more likely to need a hysterectomy, but we do know from other papers that people who are under the age 45 and those who have had painful periods are less likely to have good control of their bleeding with an ablation.
The take home message: if you have an ablation, there is a one in six chance in the next five years that you will have a hysterectomy. This doesn’t mean the procedure is good or bad, this is just data to help with decision making. Many people don’t want major surgery (meaning a hysterectomy), so a 88% chance that you won’t be dissatisfied enough that you need a hysterectomy over the next five years might seem like great odds to many. And conversely, some people might really want a one-and-done procedure, so a 12% chance of needing another procedure might seem high.
One additional point, if you have an endometrial ablation and you decide you want to take menopausal hormone therapy, you still need to take a progesterone or progestin as there is no guarantee that every spec of endometrium was removed with the procedure.
How Effective are Hormonal IUDs at Eight Years?
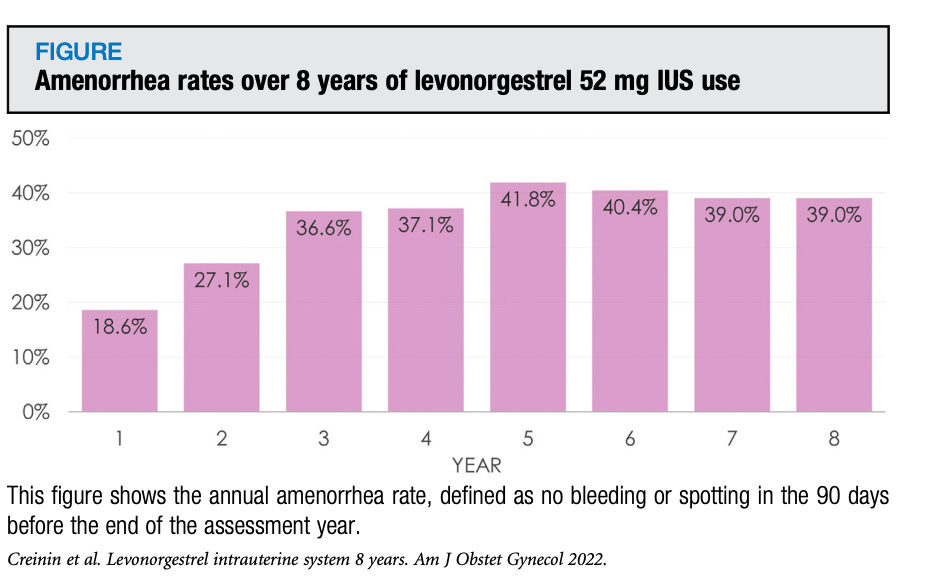
We now have eight year follow up data from ACESS IUS, which is a long-term study of people who received the Liletta IUD, a 52 mg levonorgestrel IUD (this data should also count for the Mirena). This most recent paper specifically looked at the eighth year of follow up for both pregnancy rates and bleeding profile, specifically a lack of bleeding or amenorrhea (meaning no bleeding, not even spotting). This chart caught my eye, and I wanted to share it, because who doesn’t love a good chart?
From year three onwards just over one third of people (37%-42%) using a 52 mg levonorgestrel IUD can count on not having any bleeding. For example, if you are using an IUD to control your periods and you are at year five, you can count on at least three more good years. In year 8 there were no pregnancies (in year seven the pregnancy rate was 0.25/100 woman years) and the cumulative pregnancy rate over eight years was 1.09% (this also includes ectopic pregnancies). And yes, the 52 mg levonorgestrel IUD is approved for 8 years of contraception.
Long-term data on contraception gets harder to accumulate over time, so whether we’ll eventually learn that the 52 mg IUD is good for longer than eight years remains to be seen. One reason for this difficulty is many people eventually get their IUD removed to get pregnant, so the pool of people who might get to eight years dwindles. In addition, people get lost to follow up in studies, and the longer the study, the greater the chance. And finally, people “age out” (for lack of a better phrase) of contraception studies, because investigators need to use people age 39 or younger at the start of the year. Pregnancy rates drop with age, so pregnancy rates for people who are, for example, 45 years or older can’t be translated to those who are under age 40. Obviously, the longer the duration of the study, the more likely there are to be participants who reach their 40th birthday, and hence can’t be included.
Is it really a problem to have your ovaries out before age 65?
This is about what is known as opportunistic oophorectomy, a term I hate because it sounds predatory, but it’s the concept of having your ovaries removed if you are having a hysterectomy and are close to menopause/in menopause.
Conventional teaching for several years has been that we should not remove ovaries, if we can help it, before age 65. The reasoning was that after menopause the ovaries are still hormonally active in other ways, contributing some testosterone that could be converted into estrogen, and statistical modeling suggested that when ovaries were removed before age 65 there was an increased risk of heart disease. Balancing this against the risk of ovarian cancer (which obviously decreases when the ovaries are removed) and other risks, the net effect of removing ovaries before age 65 was a shorter life expectancy. This thinking only ever applies to people with no pressing medical reason to remove their ovaries. For example, if you have a mass on your ovary that is concerning for cancer, you have ovarian cancer, or are at high risk for ovarian cancer for genetic reasons, removing your ovaries is obviously a net benefit for you and any risks related to early removal must be balanced against the significant net benefit.
However, a new look at the statistical modeling into question suggests the net negative for removing ovaries isn’t age 65, it’s actually age 50. This data is supported by a recently study from Canada that looked at more than 200,000 people and concluded that the risk of death was slightly higher when ovaries were removed before age 50 and slightly lower when removed over age 55, and maybe just barely slightly lower when removed between ages 50 and 54. It’s important to note that taking hormone therapy can mitigate the increased risk associated with removing ovaries before age 50.
These studies with statistical modeling are interesting, but obviously they can’t account for a myriad of variables such as, access to health care, family history that might drive decision making, other risk factors, and of course symptoms. Someone who is 51 and still having fairly regular periods is almost certainly going to have worse symptoms when she has her ovaries removed versus someone who is 51 and hasn’t had a period in 4 years. This is an important consideration because many people have a harder time managing with symptoms created by surgical menopause, regardless of age (although the younger you are, generally the worse the symptoms).
I’d like to point out that in the United States we remove WAY more ovaries (and do way more hysterectomies) than other countries, but we do not have a better life expectancy compared with placed where these surgeries happen less frequently. For example, in the early 2000s, a woman having a hysterectomy for non cancer-related reasons in the United States who was premenopausal had a 54% chance of having her ovaries removed, but that rate was only 30% in Australia versus 12% in Germany.
I’m not 100% convinced this kind of statistical modeling provides the whole picture, but combined with the data from the Canadian study it seems that removing ovaries over age 50 doesn’t have the negative impact on the heart that we once thought, and so if someone were having a hysterectomy at, for example, age 52 and wanted their ovaries out, there isn’t a compelling reason not to do that surgery.
DHEA in the Press
This week the Washington Post had this zinger of a headline and article, How a Renowned Fertility doctors profits from an unproven supplement (click here to read), about DHEA (dehydroepiandrosterone) supplements. It’s well worth the read. DHEA comes up a lot for menopause, so expect a post on that from me in the next few weeks.
And Finally…
I’m speaking at a menopause week called Making Over Menopause, September 19-24 at Canyon Ranch, hosted by the amazing Stacy London. You can find more about that event here. And as the publication date for my new book, Blood, gets closer I’ll keep you all updated on the tour schedule.
What’s Up Next?
I’ll be continuing the Hormone Menoverse series: next up the reasons we don’t recommend menopausal hormone therapy (meaning the red light reasons). And then we’ll tackle who can and can’t safely take menopausal hormone therapy.
If there’s something you’d like me to address in a future post, please drop it in the comments section. And if you like these shorter takes, also let me know!
References
Oderkerk TJ, Beelen P, Bukkems ALA, Van Kuijk SMJ, Sluijter HMM, van de Kar MRD, Herman MC, Bongers MY, Geomini PMAJ. Risk of Hysterectomy After Endometrial Ablation: A Systematic Review and Meta-analysis. Obstet Gynecol. 2023 Jul 1;142(1):51-60. doi: 10.1097/AOG.0000000000005223. Epub 2023 Jun 7. PMID: 37290114.
Creinin MD, Schreiber CA, Turok DK, Cwiak C, Chen BA, Olariu AI. Levonorgestrel 52 mg intrauterine system efficacy and safety through 8 years of use. Am J Obstet Gynecol. 2022 Dec;227(6):871.e1-871.e7. doi: 10.1016/j.ajog.2022.05.022. Epub 2022 May 13. Erratum in: Am J Obstet Gynecol. 2023 Jul 24;: PMID: 35569516.
Rush, Shannon K. MD; Ma, Xiuyu PhD; Newton, Michael A. PhD; Rose, Stephen L. MD. A Revised Markov Model Evaluating Oophorectomy at the Time of Hysterectomy for Benign Indication: Age 65 Years Revisited. Obstetrics & Gynecology 139(5):p 735-744, May 2022. | DOI: 10.1097/AOG.0000000000004732
Cusimano MC, Chiu M, Ferguson SE, Moineddin R, Aktar S, Liu N, et al. Association of bilateral salpingo-oophorectomy with all cause and cause specific mortality: population based cohort study. Br Med J 2021;375:e067528. doi:10.1136/bmj-2021-067528




I'd love to see more information on uterine and vaginal prolapse. Causes, things that contribute, ways to help prevent, who may be more susceptible, treatments, non-surgical options, effects if it goes untreated, etc.
I live in Canada and recently listened to an episode of the podcast 'White Coat, Black Art', where they were talking about fallopian tube removal and its effectiveness on prevention of ovarian cancer. I learned that ovarian cancer often starts in the fallopian tubes and then spreads to the ovaries and this procedure is therefore preventative. It is only performed when a woman is already undergoing another type of pelvic surgical procedure. More information/stats on that would be interesting if you've got it!
I like the short takes!