Today is International Woman’s Day, and I can’t think of a better way to honor and respect the day than to share a clinical trial that looks at testosterone and quality of life, as this topic is such a source of much misinformation online.
Remember, misinformation is the patriarchy as it robs women of agency.
Can testosterone improve the quality of life for premenopausal women with diminished ovarian reserve? That is the question a group of investigators sought to answer in a recently published double-blind, randomized, placebo-controlled trial.
What is Diminished Ovarian Reserve and Why Use Testosterone?
According to the American Society for Reproductive Medicine (ASRM), ovarian reserve is “the number of oocytes [eggs] remaining in the ovary, or oocyte quantity (oocyte number). Markers of ovarian reserve include hormone levels and sonographically measured features of the ovaries.”
Diminished ovarian reserve (DOR) is when a woman of reproductive age has regular menstrual cycles, but her response to ovarian stimulation for fertility care is reduced compared with that of women of the same age. It’s crucial to note that DOR primarily predicts poor response to fertility medications rather than absolute inability to conceive.
There is a hypothesis that testosterone might be helpful in improving the response to IVF for women with DOR. For example, animal studies have shown that androgens might amplify the ovarian response to medications used in IVF, slow follicular loss, and promote follicular growth. There is also data suggesting that androgens might increase the number of oocytes (eggs retrieved). In addition, women with diminished ovarian reserve have, on average, lower levels of testosterone.
Medically this is all interesting, but we do not know if this association with testosterone is causation or correlation. Might lower levels of testosterone for women with DOR be the result of the declining number of eggs and/or egg quality? Yes. Might the same “thing” (for lack of a better word) that leads to diminished ovarian reserve also lead to low levels of testosterone. Yes. And we also don’t know the implication of lower testosterone as there is no lower level of testosterone for women below which symptoms or medical conditions develop.
The biological rationale for testosterone supplementation during IVF is therefore a sound hypothesis, but the studies available were low quality. In fact, a 2025 meta-analysis of studies found that, when restricted to high-quality randomized controlled trials, there was no benefit of testosterone on pregnancy rate.
The next step, when there is a sound hypothesis but no robust data, is to design and implement a high-quality randomized controlled trial. And so, to evaluate a possible improved response to IVF with testosterone, a multi center randomized double blind placebo controlled trial was carried out, where women with DOR received transdermal testosterone or placebo for about 9-10 weeks as part of the preparation for their hormonal stimulation with IVF.
That’s All Very Interesting, But What Does It Have to Do with Well-Being?
The investigators built evaluating quality of life into the study design. It’s an interesting, and important decision, because diminished ovarian reserve is linked with lower quality of life and also with lower levels of testosterone. It makes sense to try to get data here too, if possible.
The investigators designed the study so the impact of testosterone on quality of life was a pre-planned secondary outcome, meaning that while they were collecting data on fertility outcomes, they were also collecting data on quality of life.
The clinical trial was conducted at eight clinics in three countries (Spain, Belgium, and Denmark). Women were randomized into two groups, one that received a 1% testosterone gel that delivered 5.5 mg of testosterone per day vs. a second that received a placebo gel. The women took the testosterone or placebo gel for their first menstrual cycle in the study and then for the next 35 days (about 9 weeks of testosterone in total).
The investigators used a scale called FertilQoL, which is a validated patient-reported outcome measure designed to assess how infertility affects a person’s quality of life. It captures the emotional, relational, and social impacts of infertility as well as experiences with fertility treatment. It is widely used in infertility research and clinical settings to measure outcomes that extend beyond pregnancy rates.
What Were the Findings?
The study was halted prematurely because testosterone did not improve IVF outcomes. A point was reached at which it would not be possible to demonstrate a statistically significant response, so the study was terminated early. Clinical pregnancy rates for testosterone were 15.7% vs 14.9% for placebo.
For the well-being aspect of the study, there was data on 106 women who received testosterone and 107 who received placebo. As expected, the testosterone levels remained low for the women on placebo (average 0.6 nmol/L or 17.3 ng/dL) and rose to the upper end of normal.just above normal for the testosterone group (average 3.2 nmol/L or 92.3 ng/dL). There was no difference in quality of life between the testosterone and placebo groups for any domain measured by the FertilQoL.
What are the Study’s Weaknesses?
The investigators used an immunoassay, which may be less accurate at low levels than mass spectrometry, so that may affect the validity of the testosterone levels, especially at the lower end.
While the FertiQoL is a validated scale, we don’t know what change represents something that is meaningful or beneficial.
The study may not have been long enough; 9 weeks is relatively short, although benefits are typically seen by 8 weeks in most studies.
The trial was powered to detect a difference in clinical pregnancy rate (the primary outcome) and was ended early, so the researchers did not enroll the target sample size. No power calculations were reported for the secondary outcome of quality of life (this is typical), but it means there may not be sufficient statistical power to make conclusions about testosterone on quality of life.
There is also an inherent limitation with evaluating secondary outcomes. The authors acknowledged the limitation, stating: “QOL was a secondary outcome in this trial, and participants were not recruited based on a low QOL.” Here are a couple of ways that a secondary outcome could introduce potential problems:
The average total FertiQoL in the study was 73-74 which is on par with the general IVF population. This is a higher score than is seen when people have depression or anxiety. It’s possible there isn’t much room to move with these results, creating a ceiling effect, making it harder to detect a treatment benefit (assuming one exists).
If participants have the full spectrum of quality of life scores (not just those with low scores), any treatment effect in a subgroup who might actually benefit gets diluted by the majority who had no room to improve or didn’t need improvement.
Is it possible the dose wasn’t high enough? The investigators kept the testosterone level at or close to the upper limit of normal. Going higher risks greater side effects, such as hair loss on the head, facial hair, voice changes, acne, and an enlarged clitoris. So this is hard to consider this a fault.
A Small Detour to Discuss How Outcomes Get Abused By Marty Makary and Some Influencers
This study brings up an important topic, secondary outcomes. A secondary outcome, as mentioned above, is pre-specified in the trial design. A questionnaire or blood test or whatever outcome is decided upon, and then the data is collected along the way along with the data for the primary objective for the study. It provides good hypothesis generating data, and it stronger when it is supported by the body of literature, but we want to be careful about drawing sweeping conclusions from secondary outcomes.
There is also something called a post-hoc analysis, meaning, the study did not specifically control for or collect data about an outcome, but after the fact, investigators sift through the data to see what they can piece together. Post hoc analyses carry a much higher risk of false positives because investigators can (consciously or unconsciously) search through the data until they find a “significant” result. This is sometimes called “p-hacking.”
A secondary outcome, while imperfect, at least demonstrates that the investigators committed to examining that endpoint before seeing the results. It is a post-hoc analysis of the DOPS study that Marty Makary and others wave around as “poof” that menopause hormone therapy prevents cardiovascular disease. It’s absurd, and I’d argue it’s misogyny, because it rests on telling women they should accept very weak, potentially extremely flawed data as science.
How Does this New Study Fit With The Body of Research?
This is the question we should be asking with every new study.
This paper is not the only randomized-placebo controlled trial to fail to show a benefit of testosterone for quality of life and/or mood. Several studies have used validated tools to measure mood and/or depression in response to testosterone:
In 2008, a 16 week randomized-double blind placebo controlled trial where 261 women ages 35 to 46 years with libido-related concerns were evaluated with three different doses of transdermal testosterone or a control group. Well-being was evaluated as well as impact on libido. There was no benefit on well-being. Quality of life/wellbeing was a secondary outcome measure.
In 2014, a study of 44 women ages 35-55 years with decrease in libido related to antidepressants were randomized to a group with a 300 mcg testosterone patch for 12 weeks or placebo group for 12 weeks. While testosterone improved libido for the women, there was no change in general well being or depression. How can testosterone impact libido and not well-being? They are different outcomes, and that is why it’s important to use validated scales so we can be sure where a benefit actually lies. Quality of life/wellbeing was a secondary outcome measure.
Also in 2014, 128 women with primary ovarian insufficiency were randomized to a 150mcg testosterone patch group or placebo group and after one year of use there was no difference in mood or wellbeing. This is the study that provides the strongest argument against testosterone as it was a 12-month study and Quality of life/wellbeing was a PRIMARY outcome.
The only data that show benefit for mood and well-being are smaller, lower-quality studies. It is important, as the science progresses, that we do not get anchored to lower-quality data simply because it supports a bias about wanting testosterone to be the answer.
Putting it All Together
I know many people will wonder, how can people feel fine when their testosterone levels are in the low normal range or not have any improvement when their testosterone levels rise from low to high normal?
Here are some things to consider, which I think are very important given the over promotion of testosterone as a menopause-fountain of youth:
Levels don’t tell the whole story. Testosterone has a rich intracrinology, meaning we don’t know what is happening inside the cell. There may be a myriad of adaptations inside the cell which we simply can’t measure, so what is going on in the blood may be meaningless.
No study has shown a testosterone level below which symptoms or diseases emerge for women. Low levels may be just fine, remember, adrenal glands continue to make testosterone. So what some doctors are calling “low” and in need of supplementation may simply be in the normal range.
Based on the data we have, it’s not likely that testosterone is helping to improve libido because it is replacing a hormone that is missing. Rather, it is likely working in the same way that most medications work, because increased libido is simply an effect of the drug.
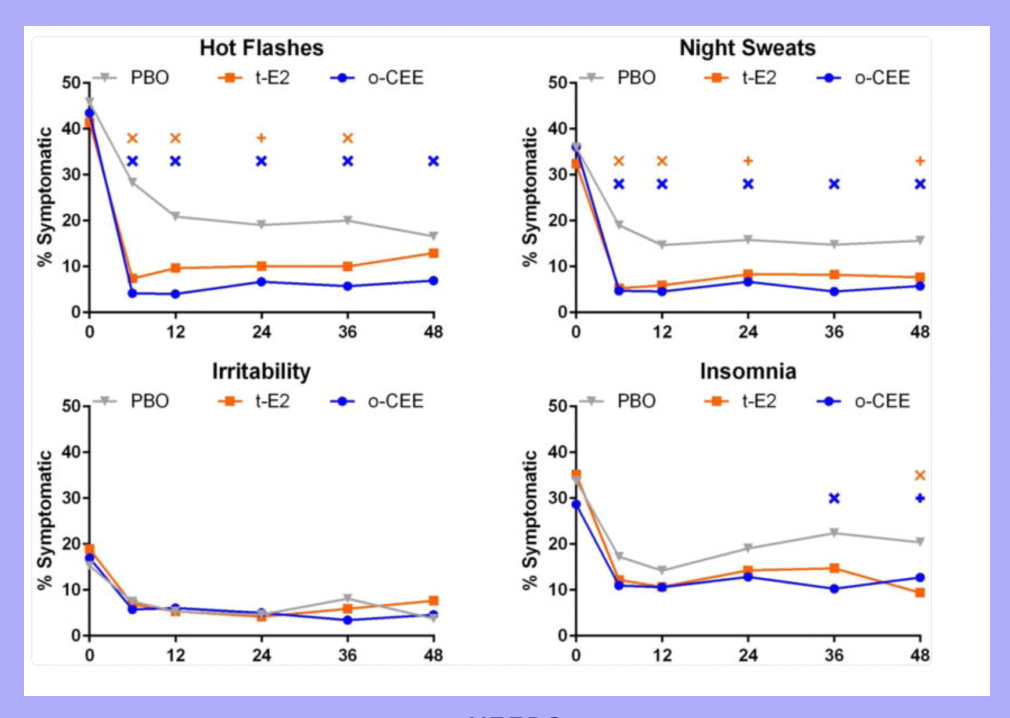
I know influencers and doctors hype testosterone online and in the office, but I don’t see these results in my practice. In fact, I do a lot of testosterone de-prescribing of people who have been on it for some time and and are clearly not getting the desired result. It’s important to remember that many medications we use in perimenopause and menopause have a very robust placebo response. I always show this slide from KEEPS when I am giving talks, as it shows the impact of both of the estrogen arms (transdermal estradiol and Premarin) as well as the placebo arm, and durable nature of the placebo response. In every metric there is drastic placebo response, and for irritability, estrogen and the placebo were indistinguishable for 48 months! About a 75% improvement!
If testosterone is essential for mood or well-being, it is reasonable to hypothesize that women with diminished ovarian reserve who generally have lower testosterone levels might benefit the most from therapy. This was a good group to study the impact of testosterone.
This study adds another piece to the body of evidence suggesting that testosterone therapy for premenopausal women (and women with primary ovarian insufficiency and those in early menopause) does not meaningfully improve overall well-being. However, it’s also important to add that we can really only have a low certainty in this conclusion, because most of the data is from secondary outcomes.
For me, the findings reinforce two important lessons in women’s health research: plausible biological mechanisms do not guarantee clinical benefit, and enthusiasm for hormone therapies should not outpace the strength of the evidence supporting them.
While the literature in the area is most secondary outcome measures (with the exception of the 12 month trial for primary ovarian insufficiency), and are not definitive, the best quality data all supports no effect for testosterone. That is why it is so irritating to see testosterone so over promoted. The data is trending to no effect, and telling women they should ignore that in the face of anecdotes is the opposite of science.
Women deserve science and an accurate representation of that science, not marketing. Anything less is the patriarchy.
References
Testing and interpreting measures of ovarian reserve: a committee opinion. ASRM
Conforti A, Carbone L, Di Girolamo R, et al. Therapeutic management in women with a diminished ovarian reserve: a systematic review and meta-analysis of randomized controlled trials. Fertil Steril. 2025
Gleicher N, Kim A, Weghofer A, et al. Hypoandrogenism is Association With Diminished Functional Ovarian Reserve. Human Reproduction. 2013.
Lin LT, Li CJ, Tsui KH. Serum Testosterone Levels Are Positively Associated With Serum Anti-Mullerian Hormone Levels in Infertile Women. Scientific Reports. 2021.
Neumann K, Kayser J, Depenbusch M, Schultze-Mosgau A, Griesinger G. Can a quality-of-life assessment assist in identifying women at risk of prematurely discontinuing IVF treatment? A prospective cohort study utilizing the FertiQoL questionnaire. Arch Gynecol Obstet. 2018
Kitchen H, Aldhouse N, Trigg A, Palencia R, Mitchell S. A review of patient-reported outcome measures to assess female infertility-related quality of life. Health Qual Life Outcomes. 2017
Davis S, Papalia M-A, Norman RJ, et al. Safety and efficacy of a testosterone metered-dose transdermal spray for treating decreased sexual satisfaction in premenopausal women. Ann Intern Med 2008
Fooladi E, Bell RJ, Jane F, Robinson PJ, Kulkarni J, Davis SR. Testosterone improves antidepressant-emergent loss of libido in women: findings from a randomized, double-blind, placebo-controlled trial. J Sex Med. 2014
Guerrieri GM, Martinez PE, Klug SP, et al. Effects of physiologic testosterone therapy on quality of life, self-esteem, and mood in women with primary ovarian insufficiency. Menopause. 2014




“plausible biological mechanisms do not guarantee clinical benefit, and enthusiasm for hormone therapies should not outpace the strength of the evidence supporting them.” 👏👏👏I want to leave this comment all over the internet on every influencer post touting testosterone, peptides or whatever the latest craze is as the panacea of wellness. Your enthusiasm and ability to turn a profit does not equal a body of evidence. I take people off testosterone all the time and don’t get me started on DHEA.
"Levels don’t tell the whole story. Testosterone has a rich intracrinology, meaning we don’t know what is happening inside the cell. There may be a myriad of adaptations inside the cell which we simply can’t measure, so what is going on in the blood many be meaningless."
YES.
I try to explain this to people and it never quite gets through. You've summarized it so well. We need to appreciate the complexity of hormone dynamics while recognizing that we have little to no basic biological research on that exact complexity during this phase of life. We don't have animal models to study menopause and... well... we science doesn't exactly have a great track for focusing on the female body or funding research that does focus on it.
Kinda feels like we're flying blind, right?